 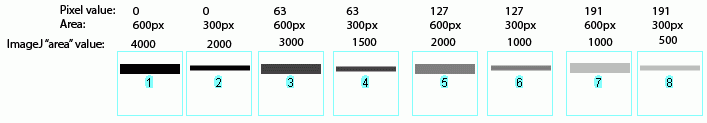
transfer, export or re-export any Bio-Rad product to Your customers or any intermediate entity in the chain of supply if any Bio-Rad product will be used in the design, development, production, stockpiling or use of missiles, chemical or biological weapons or for nuclear end uses or.sell, license, or otherwise provide or ship, directly or indirectly, any Bio-Rad product or technical data (or the direct product thereof) for export or re-export to the embargoed or restricted* countries (Cuba, Iran, *North Korea, Sudan, and *Syria) or.use any Bio-Rad product if You are located in, under the control of, or a national or resident of any embargoed or restricted* countries (Cuba, Iran, *North Korea, Sudan, and *Syria) or.You agree that, unless You receive prior authorization from the United States government, You and/or Your organization shall not: Export-Re-export Requirements and Enhanced Proliferation Control Initiative. export control laws, including but not limited to the U.S. Thanks for your input.Export of any Bio-Rad product or technical information relating to the product (i.e., manuals, memoranda, specifications, schematics, etc.) outside of the United States is subject to U.S. I’ll attach the link to the paper I’m talking about, I thought their methodology seemed pretty reliable, however I am not too far into my graduate studies so maybe I’m missing something. I’ll try out what you suggested, I am relatively new to imagej so I appreciate the suggestion and any other tips you may have. We know we have the same issue because we blot for a protein that we know has very different activity between the groups we are testing, yet when we quantify with single protein (we have used multiple to rule out biological effect) the numbers are all over the place even though we see a clear difference. 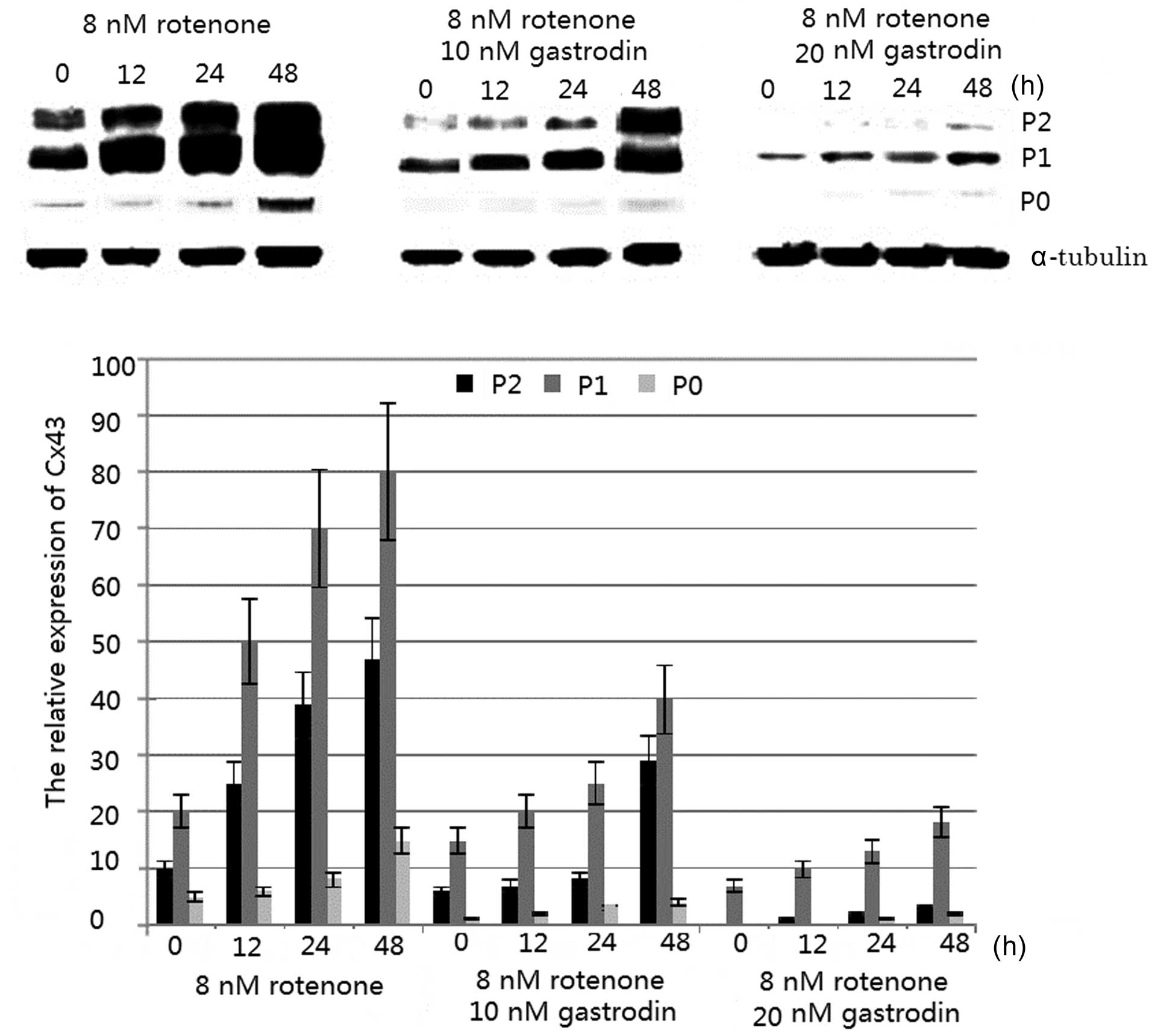
They did this by taking different aliquot of homogenates with known concentrations and assessing how well the two types of loading controls correlate to the amount of protein they know they loaded. I’ve also read literature which compares using total protein stains vs single protein loading controls and they have found that the total protein stains consistently lead to more accurate and reproducible quantification. My lab has recently found that even when our ponceau stains look homogeneous our single protein loafing controls such as GAPDH or pan actin have been highly variable after harsh stripping. ¹This subreddit is not affiliated with the creators of ImageJ or FIJI, but is simply a place to share ideas, papers, resources, and expertise - especially as relate to questions & answers posted here. Sign-up for one of the mailing lists: /Mailing_Lists It also hosts a forum for interacting with the developers.įIJI Is Just ImageJ - "a distribution of ImageJ (and ImageJ2) together with Java, Java3D and a lot of plugins organized into a coherent menu structure." is full of ImageJ development and analysis resources. Image analysis is interdisciplinary, so clearly explain field-specific terms or jargon. Clearly explain what you are trying to learn, not just the method used, to avoid the XY problem. Provide details: Be thorough in outlining the question(s) that you are trying to answer.People from the future may be stuck trying to answer the same question. Report spam or content that is hateful or off-topic.Upvote those who contribute to the discussion and provide freely of their time to assist you.Projects: Share a Link to your pet image analysis project.Research: Links to published (articles in scientific journals or in established repositories) that utilize ImageJ/FIJI for image analysis or are about image analysis.Discussions: Text posts, meant to ask about general issues relating to image analysis.Image analyst job posts are also welcome. Tips: Text or Link posts to share useful how-to tricks and discoveries on using ImageJ/FIJI.Questions which have been Solved will be marked as such. This could include algorithms, microscopy and scientific imaging, plug-ins, methods, and specific features of the software. Questions: Text posts asking about image analysis and ImageJ/FIJI.This is a an unofficial¹ forum to discuss image analysis, software features, to get help, to share ideas, and to share work done using ImageJ or FIJI. It's used worldwide, by a broad range of scientists. ImageJ is a freely available, open source image processing and analysis program using Java, on which FIJI is based. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
